Mobile Medical Applications
Solving the most complicated SaMD and connected device problems.
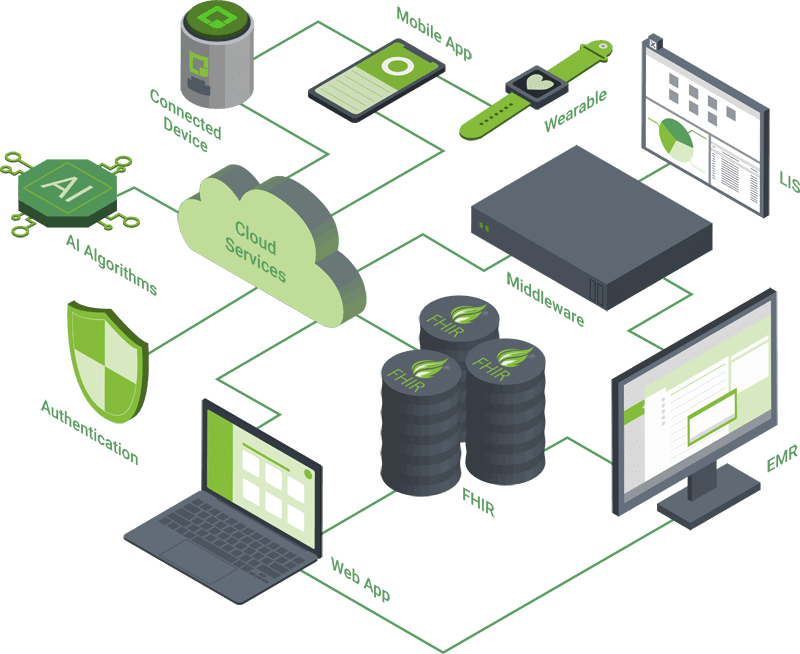
Orthogonal has developed dozens of mobile medical applications for both iOS and Android, from standalone SaMD to continuous glucose monitors to device control apps for Class III active implantables. Working with both native and cross-platform development tools, such as Flutter and React Native, we ensure support for a wide range of Bluetooth and NFC-enabled mobile devices in an ever-shifting BYOD environment.

Bluetooth, Cellular & NFC
Putting background communications
front-and-center.
Orthogonal specializes in developing mobile applications and cloud services that connect to implanted and wearable devices using Bluetooth, cellular and NFC technologies. With our extensive library of corner cases, testing methods and mitigations, we get the most out of wireless communication, maximizing reliability and speed while minimizing power consumption.

Cloud
Architecting high-performance, scalable and validated cloud environments.
Orthogonal has extensive experience applying techniques like auto-scaling by tier, self-healing and health and performance checks for Amazon Web Services, Microsoft Azure and Google Cloud Platform. That ensures that we successfully balance performance, scalability and data security with regulatory and standards compliance. As co-authors of the AAMI Consensus Report on Cloud Computing for Medical Devices, we are leading the industry on cloud validation.

Machine Learning & Artificial Intelligence
Improving device performance based
on real-world data.
Orthogonal has developed a number of FDA-cleared AI and machine learning algorithms, including solutions in cardiac monitoring, diabetes care, in vitro diagnostics and neuromodulation – and we’re involved in the ongoing development of AI-related standards. We’re experts at tuning and deploying models on a wide range of infrastructure, and have incorporated good machine learning practices for data management, model training, validation and more into our QMS.

Cybersecurity
Staying ahead of evolving threats
in an increasingly interconnected world.
Cybersecurity is an ongoing activity that is woven into Orthogonal’s product development lifecycle. Our process is compliant with the FDA’s pre- and post-market cybersecurity guidance for medical devices, as well as AAMI TIR57:2016, ISO/IEC 27001 and the NIST framework, and continues to evolve along with the SaMD ecosystem and regulatory landscape.

Web Applications
Using modern techniques to create apps that empower patients and clinicians.
Orthogonal builds modern web applications using React and Flutter – from simple dashboards, to multi-layered apps with rendering layers, domain logic and multiple types of users. We apply clean architecture principles to ensure separation of concerns, resulting in client-side applications that are robust, testable, flexible and maintainable.

FHIR & EMR Integration
Streamlining interoperability
to make solutions more powerful.
Integration of SaMD with EMR systems is a critical part of the care delivery process. Orthogonal leverages middleware solutions that provide single uniform endpoints to different EMR instances to streamline integration. We’ve built out high-performance cloud infrastructure for mobile and web apps using FHIR and SMART for seamless and secure interoperability.