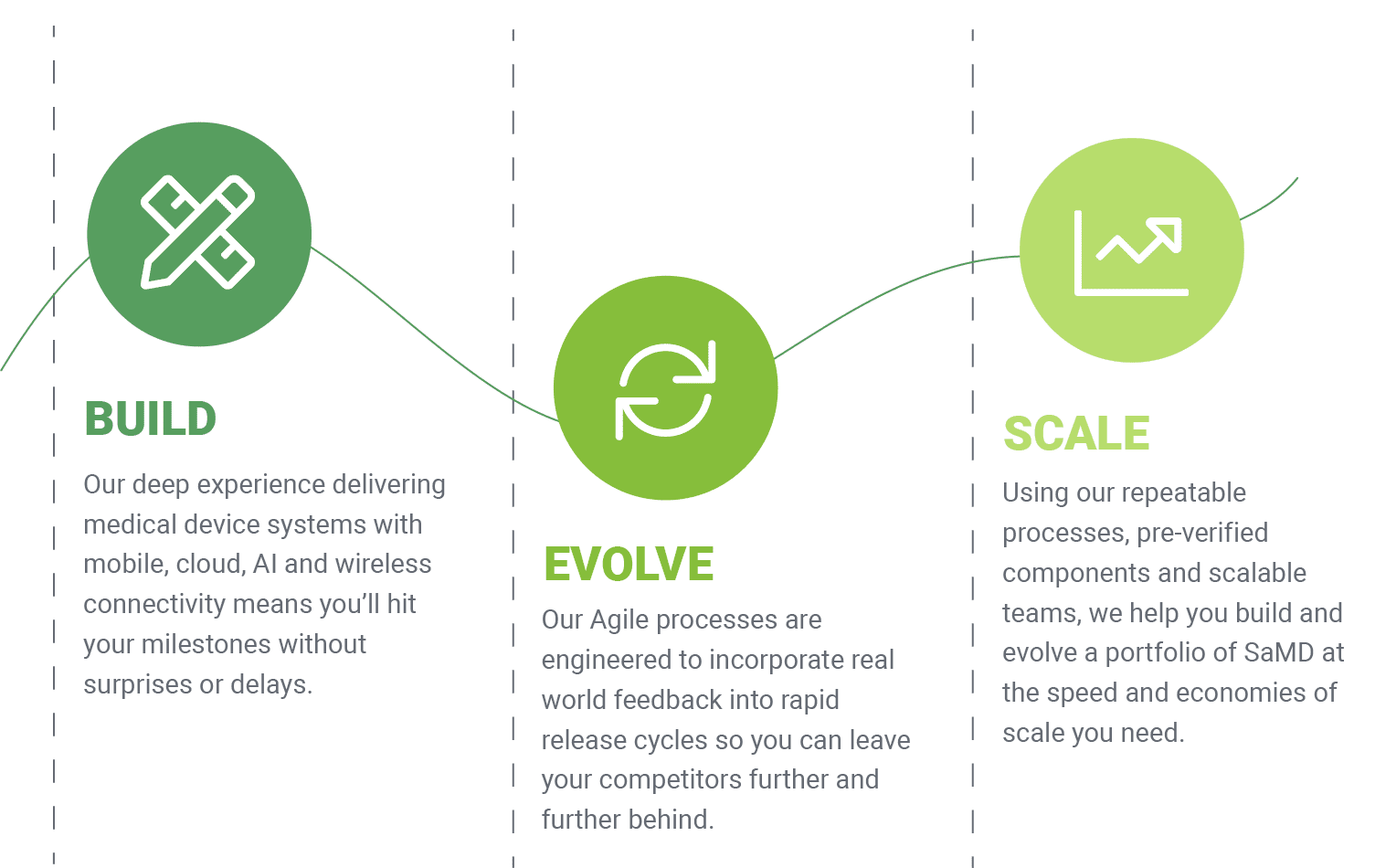
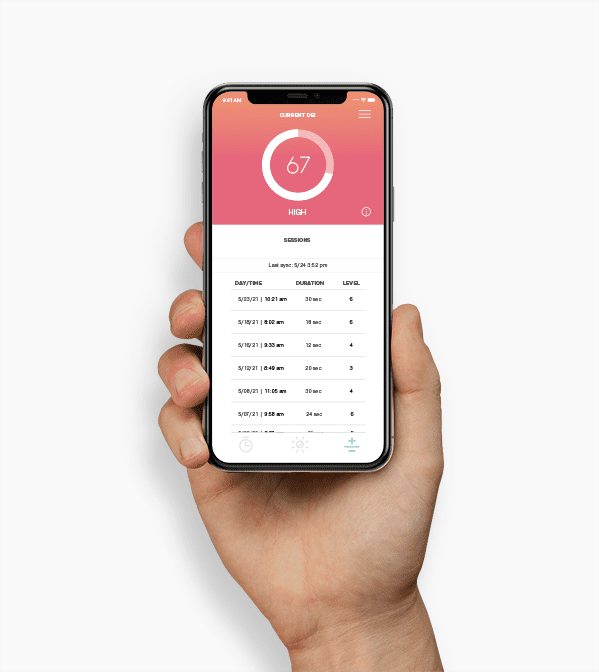
“Orthogonal’s expertise and commitment were pivotal to developing our cloud-native AI-ECG integration platform in just 7 months. Their collaborative approach, technical proficiency, and alignment with our vision helped us build a system that improves patient outcomes.”
Case Study