The Goal
Aligning your organization to reliably and predictably execute on a product vision.
SaMD Product Definition and Sizing is the planning work that allows you to reliably and predictably execute the development and launch of a SaMD product. Through it, we establish what you’ll need to realize your product vision, what you’ll have at the end of development and when your product will be ready to release. This information enables you to make important decisions on tradeoffs, allocate budget and communicate expectations to stakeholders.
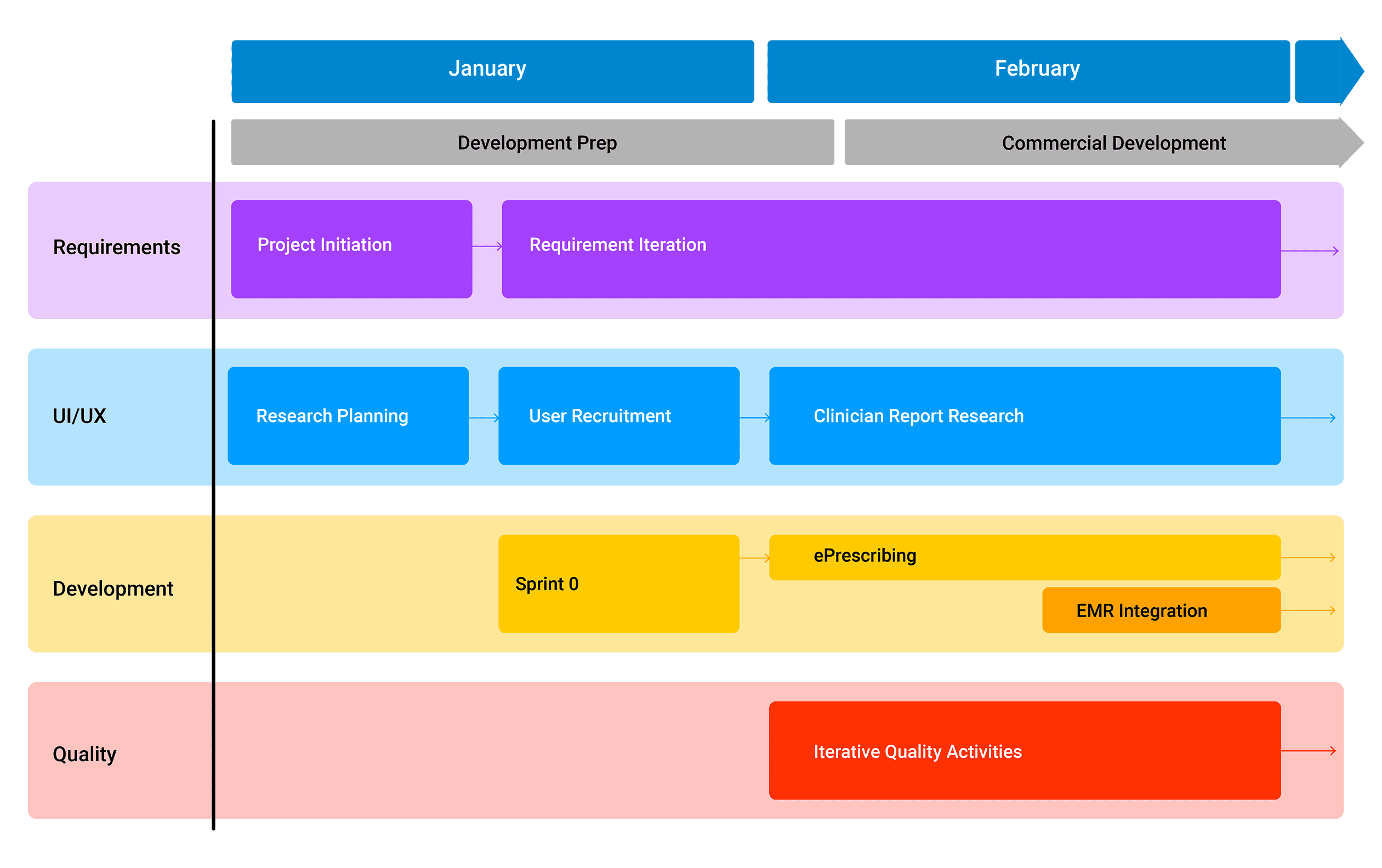
The Process & Deliverables
Generating the key components of a successful SaMD product.
Our cross-functional team of experts will join your team over a 3-6 week period in analyzing and defining the key components of a successful SaMD product. This process will generate deliverables that will guide your development efforts, encompassing functional and non-functional requirements, quality and regulatory strategy, clinical risk management, cybersecurity, UX and human factors, technical approach and verification and validation approach. We’ll use these deliverables to lay out the development timeline, expected resource allocation and costs and dependencies, as well as options for tradeoffs.
Start your SaMD Product Definition & Sizing Conversation
Take the critical first step before beginning SaMD development.
If you want to start SaMD development with a clear sense of what you’ll need, what you’ll get and when you’ll get it, then SaMD Product Definition and Sizing before committing to full development is a critical first step. For teams earlier in the journey, this often follows moving from concept to full product definition through rapid validation and prototyping. Orthogonal will help you align your organization to reliably and predictably execute on your product.

