
Article
Building Interoperability in SaMD: Tools and Best Practices
Connecting a medical device via Bluetooth to a companion app on a patient’s smartphone presents new opportunities for device manufacturers. The Orthogonal and MedSec joint webinar series on Bluetooth Low Energy for Medical Devices explores best practices for effectively implementing Bluetooth connectivity in medical devices. Expert insights from these discussions are available in the comprehensive Bluetooth for Medical Devices White Paper.

Orthogonal continues to monitor Bluetooth-enabled medical devices receiving FDA clearance. This ongoing effort tracks and analyzes new Bluetooth medical devices as they gain FDA clearance, providing insights into how these devices utilize Bluetooth technology. This updated list includes devices that have publicly announced their clearances, offering descriptions of their Bluetooth implementations.
_________________________________________________________________________
January 4th, 2024

Source: Koya Medical
Dayspring is a wearable compression system designed to be worn on the arm or leg. It is used to increase lymphatic flow, which aids in the treatment of conditions like lymphedema and edema. The device consists of a physical compression garment, a controller attached to the garment, and a mobile app. The treatment provided by the garment can be customized through a mobile app connected to the controller over Bluetooth. Patients can also adjust settings and view their treatment history on the app.
January 18th, 2024

Source: NuvoAir
NuvoAir’s Air Next is a handheld spirometer that performs basic lung function and spirometry testing for use in hospitals, clinical settings and the home. When patients breathe into the device, a digital infrared interruption software counts the number of rotations and converts that measurement into airflow measured in liters per second. Test results are shared with patients over Bluetooth on the Air Next app.
January 31, 2024

Source: Masimo
The MightySat OTC from Masimo is a fingertip pulse oximeter intended for medical use and available without a prescription. Patients place a finger inside the device to measure their functional oxygen saturation of arterial hemoglobin and pulse rate. The device includes Bluetooth technology for wireless transfer of patient data to the Masimo Personal Health app.
February 4, 2024

Source: X-trodes
The X-trodes System M is a connected medical device system that monitors and records a variety of physiological signals which are then sent via Bluetooth to a cloud server. The device consists of flexible patches known as Smart Skin that when worn can acquire medical-grade data while a patient goes about their day. Among the signals it records are electroencephalograms (EEG), electrocardiograms (ECG) and electromyograms (EMG).
February 15th, 2024

Source: Downloadable PDF on Neuro Spinal Innovation site
The SONIK MONARK 100 from Neuro Spinal Innovation is a medical device that aids in the management of chronic back pain and encourages spinal realignment. The device delivers controlled, low-frequency impulses through a stylus tip to the affected site. Treatment data is sent over Bluetooth to a connected computer.
February 15th, 2024

Source: Empatica
EpiMonitor by Empatica is a medical device system for monitoring seizures, consisting of a wearable EmbracePlus device and a companion EpiMonitor smartphone app. The wearable device senses electrodermal activity and motion data and detects potential tonic-clonic seizures in patients with or at risk of having epilepsy. If a seizure event is detected, the wearable device sends a notification over Bluetooth to the smartphone app, alerting the patient and their caregivers. Data collected by the device is also shared from the app with a cloud database.
March 5th, 2024
Source: Dexcom
Stelo is an integrated Continuous Glucose Monitor (CGM) available over the counter to patients with Type 2 diabetes who are not on insulin. Like other CGMs, Stelo continuously measures, analyzes and records glucose levels, as well as shares data over Bluetooth to a companion smartphone app. Unlike other CGMs, including Dexcom’s G7 platform, Stelo is not interoperable with insulin pumps, nor does it have the ability to suggest insulin dosages, as the device is intended for patients not on insulin.

March 15th, 2024
Source: Otodynamics
Otoport Advance is a handheld device used to non-invasively diagnose hearing loss in children and adults via Transient Evoked Otoacoustic Emissions (TEOAE) and Distortion Product Otoacoustic Emissions (DPOAE). Data from the Otoport Advance can be transmitted over Bluetooth to Otodynamics’s Otolink PC patient data management software suite.
April 10th, 2024

Source: Lapsi Health
The Keikku Electronic Stethoscope from Lapsi Health amplifies, filters and transmits listening data of a patient’s heart, lungs, bowels, arteries and veins to a companion smartphone app over Bluetooth. The Keikku app provides data annotation, visualization and audio playback, as well as sharing features so that the device can remotely monitor patients and serve as an input in telemedicine. The device is intended for use both in clinical settings and at home by patients.
April 19th, 2024
Source: Edan
The iX Series of patient monitors from Edan Instruments perform long-term continuous monitoring of a patient’s physiological parameters, including respiration rate, temperature, oxygen saturation, blood pressure and more. The device can transmit patient data over Bluetooth to Edan’s e-Link platform.
April 29th, 2024
Source: Transtek
The Transtek Blood Pressure Monitor measures systolic pressure, diastolic pressure and pulse rate using an inflatable cuff wrapped around the upper arm. Readings from the device sync over Bluetooth with the Transtek Health app, as well as a cloud database.
May 28th, 2024

Source: Clario
The Clario SpiroSphere is a compact spirometry device used to measure a patient’s inspiratory and expiratory lung function. With this 2024 filing, the device is now able to take 12-lead ECGs by connecting via Bluetooth to a compatible portable ECG device, the Corscience COR12.
May 29th, 2024
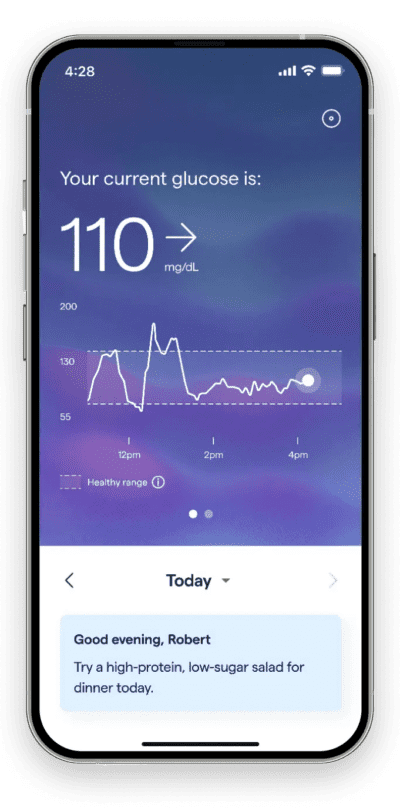
Source: Abbott
Abbott’s Lingo is an integrated CGM available over the counter, designed to monitor blood glucose levels of patients with Type 2 diabetes who do not use insulin. This device appears to compete directly with the Dexcom Stelo, as they share the same indication for use. The Lingo sensor worn on the body connects over bluetooth to a companion Lingo app, allowing patients to view their real-time glucose values, trends and graphs.
May 7, 2024

Source: Kestra Medical Technologies
The ASSURE Wearable ECG is a continuous cardiac monitoring device for adults who previously used the ASSURE Wearable Cardioverter Defibrillator system. It includes a Monitor Cable Assembly and a SensorFit™ Garment with ECG electrodes. Using Bluetooth Low Energy, it transmits ECG data to a mobile app, enabling real-time, wireless monitoring and remote access for healthcare providers.
May 28, 2024

Source: Sibel Health
The ANNE Limb is a wearable medical device for continuous vital sign monitoring in patients 12 and older. It measures SpO2, pulse rate, and skin temperature. Using Bluetooth, it transmits real-time data to the Sibel SDK, which can be integrated into various applications for display and storage.
May 29, 2024
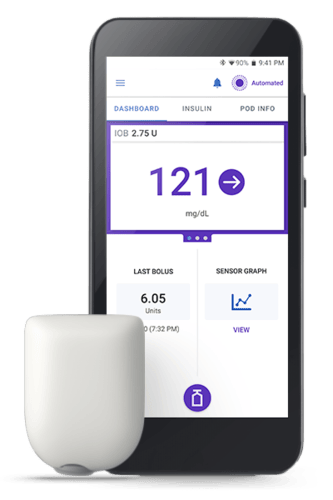
Source: Insulet
SmartAdjust™ is a software-only medical device for type 1 diabetes management. It’s an interoperable automated glycemic controller that works with compatible iCGMs and ACE pumps to automatically adjust insulin delivery based on current and predicted glucose values. The device uses Bluetooth Low Energy (BLE) for component communication.
June 7, 2024
Source: Abbott
The Libre Rio Continuous Glucose Monitoring System is an over-the-counter iCGM for adults 18+, designed for non-insulin use. It includes a sensor worn on the upper arm and a smartphone app, connected via Bluetooth Low Energy (BLE) for automatic, scan-free data transfer.
June 21, 2024

Source: Tactile Medical
The Nimbl is a portable compression device for managing edema, lymphedema, venous insufficiency, and wound healing. It uses Bluetooth to connect to a smartphone app for controlling settings, logging sessions, and sharing data with healthcare providers.
July 10, 2024
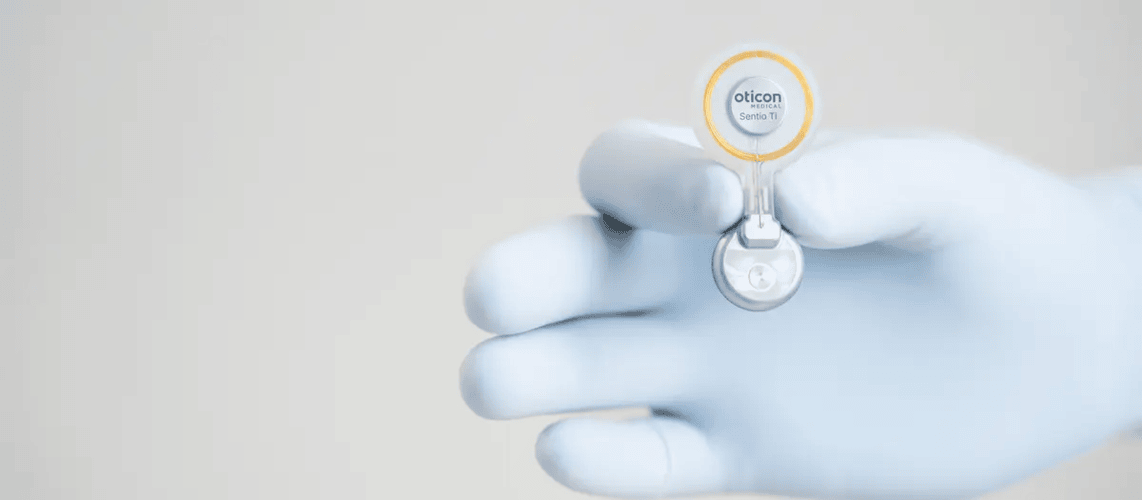
Source: Oticon Medical
The Sentio Active Bone Conduction Hearing System, with the Sentio Ti Implant and Sentio 1 Mini processor, aids patients with hearing loss or single-sided deafness. The Sentio 1 Mini uses Bluetooth Low Energy (BLE) for wireless communication, enabling volume adjustments, audio streaming, and remote fitting via the Oticon Companion app.
August 8, 2024
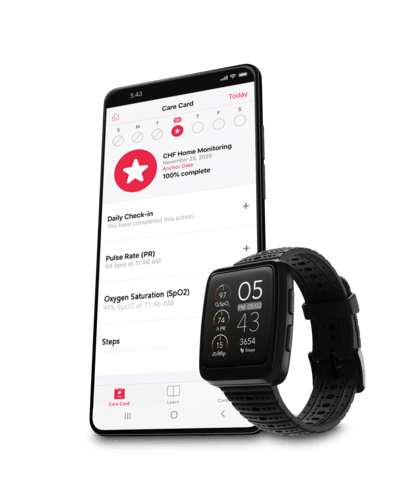
Source: Masimo
The Masimo W1 is a wrist-worn device for spot-checking vital signs, measuring heart rate, SpO2, and pulse rate. Designed for adults, it uses Bluetooth Low Energy (BLE) to transmit data to smart devices, enabling health tracking and efficient data transfer for personalized care.
August 21, 2024

Source: Nalu Medical
The Nalu Neurostimulation System is an implantable device for managing chronic pain from peripheral nerves in adults. It uses Bluetooth Low Energy (BLE) for wireless control via a Therapy Disc, secure data transmission, and remote management by healthcare providers through a Clinician Programmer Application. Patients can manage their therapy with a smartphone Remote Control Application to select options and control stimulation.
August 22, 2024
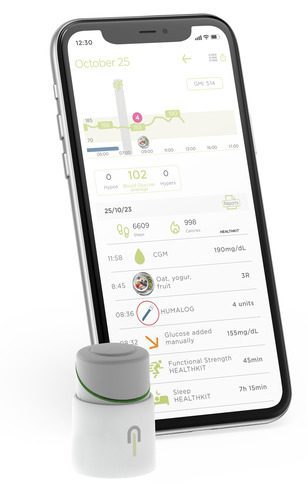
Source: Insulcloud
The INSULCLOCK® v2.0 PRO is a reusable transmitter for insulin pens, using Bluetooth to track and transfer dose data to smartphones, improving diabetes management and enabling remote monitoring by healthcare providers.
August 28, 2024

Source: OxiWear
The OxiWear System is an ear-worn device for non-invasive SpO2 and pulse rate monitoring. It uses Bluetooth to communicate with a mobile app, enabling real-time data transfer, continuous health monitoring, and access to historical trends. The app provides live updates and stores patient information, which can be shared with healthcare providers for analysis and care management.
August 30, 2024

Source: Vave Health
The Vave Wireless Ultrasound System is a portable device for diagnostic ultrasound imaging and fluid flow analysis. It features a handheld probe that wirelessly transmits images to mobile devices via Bluetooth, allowing healthcare professionals to view real-time results on smartphones or tablets. This enhances mobility in various settings and supports remote monitoring and data logging for effective patient care.
September 4, 2024

Source: Modular Medical
The Modular Medical MODD1 Insulin Delivery System manages diabetes in adults needing insulin, combining a reusable programmable pump and disposable cartridge for subcutaneous delivery. It connects via Bluetooth to the MMI App, enabling users to program basal schedules and view pump data.
September 16, 2024

Source: Senseonics
The Eversense 365 Continuous Glucose Monitoring (CGM) System is a long-term implantable device designed to measure glucose levels in people with diabetes for up to 1 year. It consists of three main components: a small sensor inserted under the skin, a removable smart transmitter worn over the sensor, and a mobile app for displaying glucose data. The system uses fluorescence technology to measure glucose levels and Bluetooth Low Energy (BLE) to transmit data from the transmitter to the mobile app.
October 31, 2024
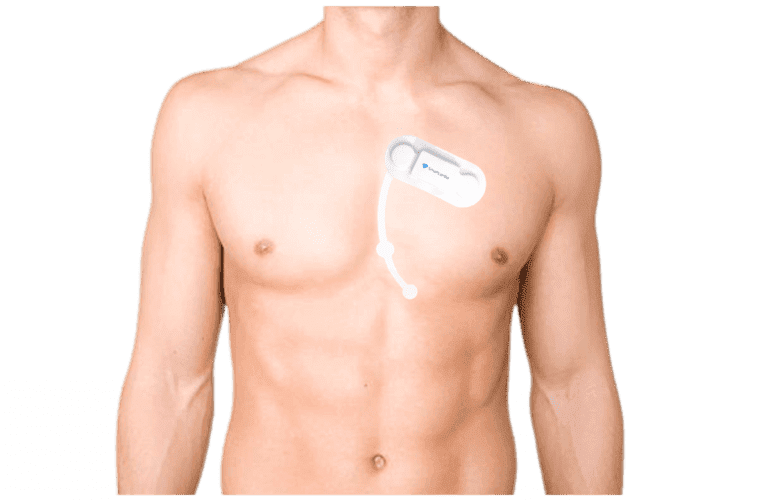
Source: SmartCardia
The SmartCardia 7L Platform MCT is a wearable device for continuous ECG monitoring, comprising a disposable 7L Patch, reusable 7L Sensor, SmartCardia Phone, and cloud service. It uses Bluetooth to wirelessly transmit heart rate and ECG data from the Sensor/Patch to the Phone for real-time monitoring and symptom recording, enhancing usability and patient comfort.
November 8, 2024

Source: Fourth Frontier
The Frontier X Plus is an advanced ambulatory monitoring device designed to record, store, and transfer single-channel electrocardiogram (ECG) rhythms for comprehensive cardiac monitoring and evaluation. This innovative medical device utilizes Bluetooth Low Energy (BLE) technology to seamlessly transmit ECG data from the device to a companion mobile application.
November 25, 2024

Source: Aluna
Aluna 2 is a portable peak flow meter for monitoring lung function, measuring FEV1 and PEF. It uses Bluetooth Low Energy (BLE) to wirelessly transmit data to a companion mobile app, enabling seamless data transfer and real-time monitoring of respiratory health without manual recording.
November 29, 2024

Source: Future
The Movano Health Evie Med Ring is a medical device designed to monitor vital health parameters. This wireless, non-invasive pulse oximeter is worn on the finger and uses Bluetooth Low Energy (BLE) technology to transmit data to a companion mobile app.
Looking for a partner to develop your Bluetooth-enabled medical device?
Our expertise in BLE for medical devices and extensive library of edge cases, testing methods and mitigations will help you get the most out of Bluetooth communication for connected medical devices.

Related Posts

Article
Building Interoperability in SaMD: Tools and Best Practices
Article
Leveraging AI/ML in SaMD Development: Benefits and Challenges
Article
ISO 13485 Compliance Checklist for SaMD Development

Article
How to Conduct Post-Market Surveillance for SaMD