
Article
What Actually Works in MedTech, According to Industry Leaders
White Paper
The tragedy of COVID-19 has opened up a rare window of opportunity for innovators and leaders to have a considerable impact on healthcare costs and outcomes.
In this white paper Orthogonal shares advice based on what we see in the market for how firms can take advantage of this window to make huge gains with great digital health solutions, including connected medical devices, Software as a Medical Device (SaMD) and Digital Therapeutics (DTx).

In this whitepaper:
Related Posts

Article
What Actually Works in MedTech, According to Industry Leaders

Article
The Future of MedTech: Insights from Industry Leaders

Article
Studying Compliance Burden to Improve SaMD Development
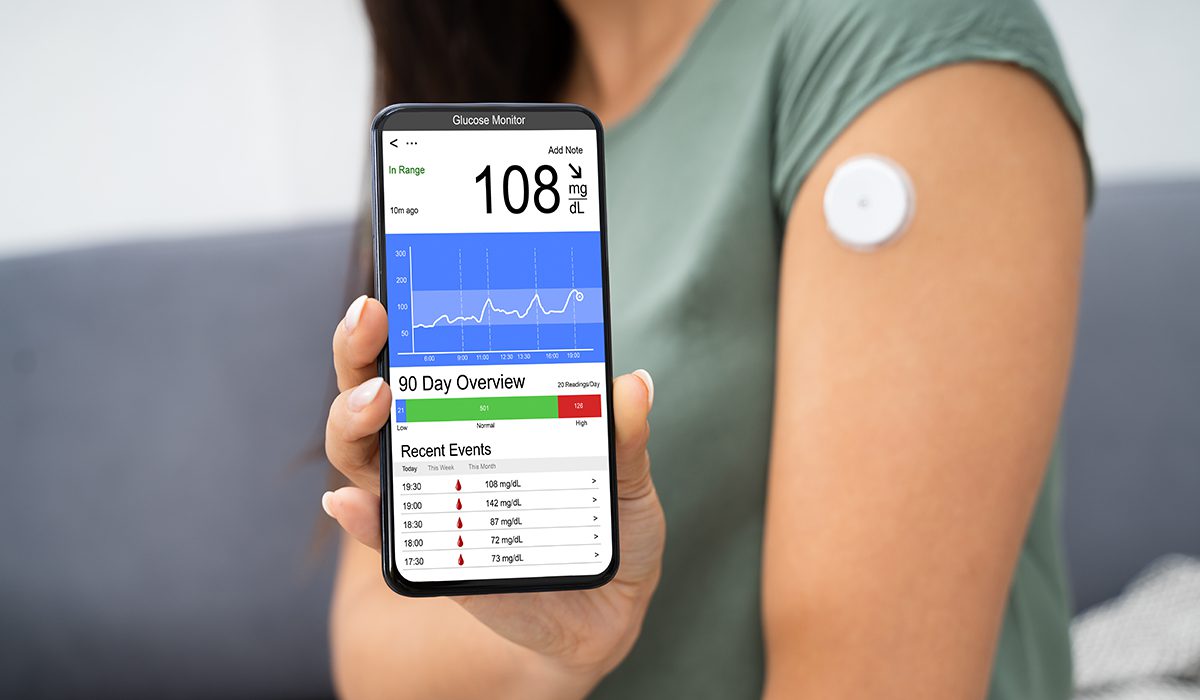
Article
Lifestyle Integration for Medical Devices