
Article
Roundtable Chapter 2 – The One With the Bottleneck at the Finish Line
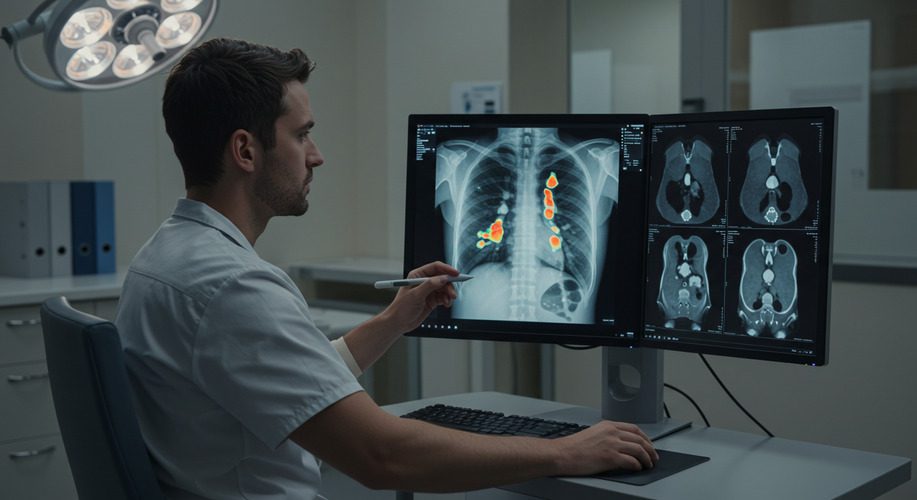
Launching Software as a Medical Device (SaMD) is a complex process that requires careful planning, regulatory adherence, and innovative execution. Examining successful SaMD launches offers valuable insights into best practices and strategies that you can apply to your own projects. This case study highlights two exemplary SaMD launches and the lessons learned from their success.
A MedTech startup developed an AI-powered imaging tool designed to detect early-stage lung cancer in radiology scans. The solution integrated seamlessly with existing hospital systems and provided real-time diagnostic support to radiologists.
Related: Building Interoperability in SaMD: Tools and Best Practices
A global MedTech company launched a remote monitoring platform for chronic heart failure patients. The SaMD used IoT-enabled wearable devices to track patient vitals and provide real-time alerts to clinicians.
Related: How to Use Real-World Evidence (RWE) in SaMD Validation
Related: How to Conduct Post-Market Surveillance for SaMD (Advanced Guide)
The success of a SaMD launch hinges on strategic planning, stakeholder collaboration, and a focus on user-centric design. By examining these case studies and implementing the lessons learned, you can streamline your own SaMD launch process and achieve impactful results.
For further insights, explore related articles:
Apply these strategies to ensure your SaMD launch not only meets regulatory requirements but also exceeds user expectations and drives meaningful outcomes in healthcare innovation.
Related Posts

Article
Roundtable Chapter 2 – The One With the Bottleneck at the Finish Line

Article
Roundtable Chapter 1 – The One Where the Device Was Only Half the Story

Article
Roundtable Prologue – The One Where the Algorithm Couldn’t Sell Itself

Article
Managing Emerging Risks in SaMD: Strategies for 2025 and Beyond