
Article
MedTech Teams Should Stop Paying for Compliance Twice
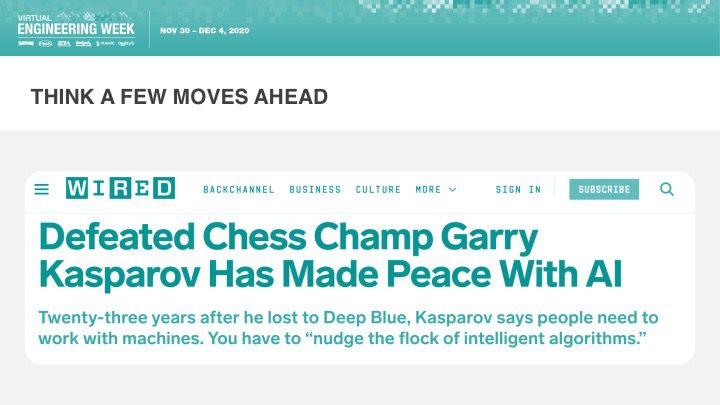
Orthogonal was excited that Bernhard Kappe, our CEO and Founder, spoke at Virtual Engineering Week on Friday, December 4th, 2020 on the topic of AI & Machine Learning in Medical Devices: It’s Getting Better All the Time. This talk was hosted by Informa and their Medical Device + Diagnostic Industry (MD+DI) media property. (Shout outs to Laurie Lehmann and Naomi Price for setting this up and guiding us through the process.)
If you missed the talk and want to check it out, or you did catch the talk and want more or want to share it, you have a few options:




















Related Posts

Article
MedTech Teams Should Stop Paying for Compliance Twice

Article
Beyond the Device: Where MedTech Digital Ecosystems Are Starting to Pay Off

Article
Why MedTech Teams Slow Down as Software Grows and What Better Architecture Looks Like

Article
What Actually Works in MedTech, According to Industry Leaders